Key Takeaways
- PAM finishes many peptide signals that control appetite, stress and blood sugar.
- Copper and vitamin C are required for normal PAM enzyme activity.
- Lower PAM activity has been linked with diabetes risk and pituitary changes.
- Nutrient dense animal foods can support the raw inputs PAM needs.
- Frequent snacking and processed low fat eating can work against hormone balance.
PAM Basics
What PAM Does
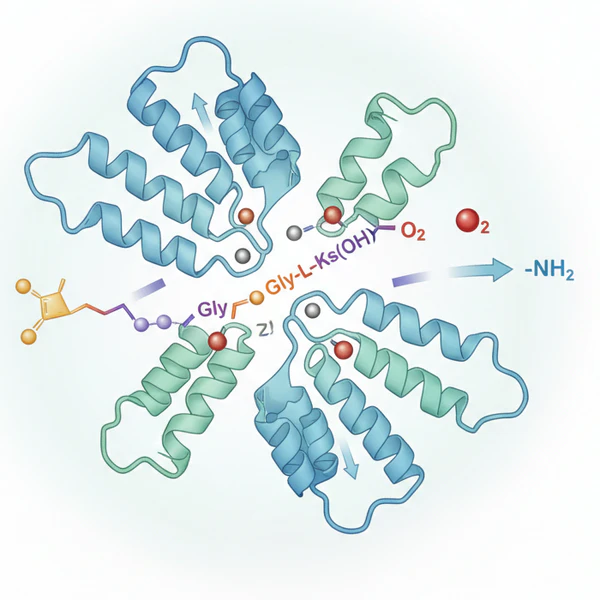
PAM gives many peptide hormones and neuropeptides their active final form. The full name is peptidylglycine alpha amidating monooxygenase. It adds a small chemical group called an amide to the tail end of certain peptides. That last step can decide whether a signal is strong enough to do its job in the brain, gut, pancreas or pituitary (1, 2). Cells use PAM near the end of peptide production. A peptide may be made in full length form yet still need that finishing step before it works well. Researchers have described PAM as a central part of peptide maturation because many signals lose potency when amidation is poor (1, 3).
PAM also does more than one narrow job inside secretory cells. Work on the enzyme has shown links with peptide processing, granule trafficking and the internal handling of signaling molecules before release. A problem in PAM can ripple across several pathways at once because one enzyme sits upstream of many peptide signals (3, 4).
Copper & Vitamin C
Copper and vitamin C are required for PAM to work. Copper helps drive the oxygen based reaction while vitamin C supports the enzyme chemistry that keeps the cycle moving. When either input is lacking, amidation can slow down and peptide signaling can weaken (2, 5, 6).
Mineral balance deserves much more attention than it gets in routine diet advice. A person can eat enough calories yet still fall short on the nutrients an enzyme needs. Diets heavy in refined carbohydrates, seed oils and fortified low fat products often crowd out the animal foods that naturally carry copper, protein and other raw materials for hormone function.
Copper rich foods include liver, oysters and other shellfish. Vitamin C can come from whole food sources such as acerola, amla or camu camu when fruit intake is modest. A nutrient dense diet gives the enzyme real support instead of relying on processed products with added vitamins.
Where PAM Works
Brain & Gut
PAM is active in neuroendocrine cells that act like a blend of nerve tissue and hormone tissue. Those cells help regulate hunger, satiety, stress response, pain signaling and gut movement. Amidated peptides are common in those systems, so PAM helps shape everyday physiology from meal timing to stress tolerance (3, 5).
The brain relies on precise signaling rather than rough estimates. A small drop in peptide quality can affect how strongly a message is received. Research on PAM has linked it with broad neuroendocrine function rather than one isolated messenger, which helps explain why the effects can look wide ranging in real life (3, 6).
Pancreas & Pituitary
PAM is present in the human endocrine pancreas and in the pituitary gland. Those tissues regulate blood sugar, growth, thyroid output, fertility and stress hormones. The location fits the clinical picture because altered PAM function has shown up in both metabolic and endocrine disorders (7, 8).
The pancreas depends on tightly timed peptide release around meals. The pituitary sends signals that direct several other glands. Poor peptide finishing in either site can disturb the rhythm and strength of those messages, which can show up as unstable energy, altered glucose handling or abnormal hormone output over time (1, 7, 8).
Plasma amidation activity has also been measured in human studies, which shows that peptide processing is not just a cell culture concept. Earlier work linked circulating amidation activity with gastrin processing and with tissue activity in humans, adding support for PAM related physiology outside narrow lab settings.
Health Effects
Blood Sugar Control
Lower PAM activity has been linked with impaired insulin release and higher type 2 diabetes risk. Human genetic work found PAM risk alleles that reduced enzyme activity through lower expression or weaker catalytic function. The result was poorer beta cell performance, which gives a direct route from gene variation to weaker glucose control (9).
Newer human work tied PAM deficiency to sarcopenic diabetes, a state with poor glucose control plus low muscle mass. Muscle is a major site for glucose disposal, so that combination can be especially harmful. The study suggests that weak peptide processing may affect body composition as well as insulin signaling (10).
Another study reported that PAM related diabetes risk alleles influenced GLP 1 levels and response to GLP 1 receptor agonists. The finding points again to altered peptide hormone handling rather than a single blood sugar defect in isolation. Peptide biology sits close to the center of metabolic control (11).
Frequent carbohydrate feeding can add stress to an already strained system. A lower carbohydrate animal based diet can reduce the number of glucose spikes a person has to manage each day. Fewer eating events often help create clearer hormone pulses as well, especially when meals contain eggs, ruminant meat, shellfish and natural animal fat.
Pituitary & Pressure
Loss of function PAM variants have been found more often in people with pituitary hormone excess. Studies have linked PAM variation with pituitary hypersecretion, prolactinomas, cyclical Cushing disease and thyrotroph tumors. Older tissue work also found increased PAM expression in pituitary tumors, which supports a real biological link rather than a loose coincidence (12, 13, 8).
Blood pressure may also be affected in some people. One Korean population study found an association between a PAM gene variant and hypertension. Vessel tone and fluid balance depend on peptide signaling, so a link with PAM is biologically plausible even though blood pressure always has several causes (14, 3).
Mainstream discussions often reduce these problems to one lab value or one drug category. Enzyme function, nutrient status, meal rhythm and tissue signaling deserve equal attention when the problem begins much earlier in the chain.
Daily Support
Meals & Nutrients
A high fat animal based diet gives PAM better raw material support than a processed low fat diet. Ruminant meat, eggs, liver, shellfish and full fat dairy for those who tolerate it can supply protein, copper, vitamin A and other cofactors in forms the body handles well. Ultra processed foods and fortified products tend to add noise without delivering the same nutrient density.
One to three meals a day often works better than constant grazing. Clear gaps between meals can steady insulin and gut signaling while giving the nervous system quieter intervals. Breakfast could be eggs cooked in butter with beef. A later meal could be lamb with broth. Another day could include sardines, steak or oysters to improve mineral intake.
Cod liver oil from Rosita Real Foods or Jigsaw Health can suit some people who need extra support. Magnesium glycinate, threonate, malate, taurate or chloride may help when intake is low. Whole food vitamin C, trace minerals, taurine and creatine monohydrate are other options that fit a nutrient focused approach.
Copper balance should be respected rather than ignored. Liver once or twice a week and shellfish on a regular schedule can cover much of the need through food. A diet based on lean chicken breast, cereal, low fat yogurt and snack bars pushes the body in the other direction by lowering copper rich foods while raising processed carbohydrate intake.
What To Avoid
Seed oils, ultra processed foods and refined grain products can work against hormone stability. Many plant foods also carry antinutrients and defense chemicals that reduce mineral availability or irritate digestion in susceptible people. Grains and many high oxalate or high lectin foods are poor choices when the goal is better nutrient density and steadier endocrine function.
Low fat eating is a common trap. Animal fat supports satiety and helps people stop chasing snacks every few hours. Cholesterol also deserves a calmer discussion than it usually gets. The body uses cholesterol as a raw material for normal physiology, and routine fear around it often pushes people toward foods that are less nourishing.
Before changing your diet, supplements or health routine, talk with a licensed healthcare professional. For any health concerns or questions about a medical condition, get guidance from a physician or another appropriately trained clinician.
FAQs
What is PAM enzyme?
PAM is an enzyme that finishes certain peptide hormones and neuropeptides. It adds an amide group to the end of those peptides so they can send a stronger signal.
Why does PAM affect hormones?
Many peptide hormones need amidation before they work well. PAM performs that final step, so lower activity can weaken hormone signaling.
Which body systems rely on PAM?
The brain, gut, pancreas and pituitary rely on PAM. Those tissues use many peptide signals to regulate appetite, stress, blood sugar and hormone release.
What nutrients support PAM activity?
Copper and vitamin C are the main known helpers for PAM. Nutrient dense animal foods are useful because they supply copper, protein and other key inputs.
Can PAM problems affect health in daily life?
Research has linked PAM changes with diabetes risk, low muscle mass, pituitary hormone excess and possible blood pressure effects. The exact impact can differ from person to person.
Research
Eipper, B.A., Stoffers, D.A. and Mains, R.E. (1992) ‘The biosynthesis of neuropeptides: peptide alpha amidation’, Annual Review of Neuroscience, 15, pp. 57 to 85. Available at: https://pubmed.ncbi.nlm.nih.gov/1575450/
Prigge, S.T., Mains, R.E., Eipper, B.A. and Amzel, L.M. (2000) ‘New insights into copper monooxygenases and peptide amidation: structure, mechanism and function’, Cellular and Molecular Life Sciences, 57(8 to 9), pp. 1236 to 1259. Available at: https://pubmed.ncbi.nlm.nih.gov/11028916/
Bäck, N., Mains, R.E. and Eipper, B.A. (2022) ‘PAM: diverse roles in neuroendocrine cells, cardiomyocytes, and green algae’, FEBS Journal, 289(15), pp. 4470 to 4496. Available at: https://pubmed.ncbi.nlm.nih.gov/34089560/
Eipper, B.A. et al. (1993) ‘Peptidylglycine alpha amidating monooxygenase: a multifunctional protein with catalytic, processing, and routing domains’, Protein Science, 2(4), pp. 489 to 497. Available at: https://pubmed.ncbi.nlm.nih.gov/8518727/
Kumar, D., Mains, R.E. and Eipper, B.A. (2016) ‘60 YEARS OF POMC: From POMC and α MSH to PAM, molecular oxygen, copper, and vitamin C’, Journal of Molecular Endocrinology, 56(4), pp. T63 to T76. Available at: https://pubmed.ncbi.nlm.nih.gov/26667899/
Bousquet-Moore, D., Mains, R.E. and Eipper, B.A. (2010) ‘Peptidylgycine α amidating monooxygenase and copper: a gene nutrient interaction critical to nervous system function’, Journal of Neuroscience Research, 88(12), pp. 2535 to 2545. Available at: https://pubmed.ncbi.nlm.nih.gov/20648645/
Martínez, A. et al. (1993) ‘Immunocytochemical localization of peptidylglycine alpha amidating monooxygenase enzymes in human endocrine pancreas’, The Journal of Histochemistry and Cytochemistry, 41(3), pp. 375 to 380. Available at: https://pubmed.ncbi.nlm.nih.gov/8094086/
Steel, J.H. et al. (1994) ‘Peptidylglycine alpha amidating monooxygenase immunoreactivity and messenger RNA in human pituitary and increased expression in pituitary tumours’, Cell and Tissue Research, 276(1), pp. 197 to 207. Available at: https://pubmed.ncbi.nlm.nih.gov/8187161/
Thomsen, S.K. et al. (2018) ‘Type 2 diabetes risk alleles in PAM impact insulin release from human pancreatic beta cells’, Nature Genetics, 50(8), pp. 1122 to 1131. Available at: https://pubmed.ncbi.nlm.nih.gov/30054598/
Giontella, A. et al. (2025) ‘Deficiency of Peptidylglycine alpha amidating Monooxygenase, a cause of sarcopenic diabetes mellitus’, The Journal of Clinical Endocrinology & Metabolism, 110(3), pp. 820 to 829. Available at: https://pubmed.ncbi.nlm.nih.gov/39137152/
Umapathysivam, M.M. et al. (2026) ‘Type 2 diabetes risk alleles in peptidyl glycine alpha amidating monooxygenase influence GLP 1 levels and response to GLP 1 receptor agonists’, Genome Medicine, 18, article 40. Available at: https://pubmed.ncbi.nlm.nih.gov/41906117/
Trivellin, G. et al. (2023) ‘Germline loss of function PAM variants are enriched in subjects with pituitary hypersecretion’, Frontiers in Endocrinology, 14, article 1166076. Available at: https://pubmed.ncbi.nlm.nih.gov/37388215/
De Sousa, S.M.C. et al. (2023) ‘PAM variants in patients with thyrotrophinomas, cyclical Cushing’s disease and prolactinomas’, Frontiers in Endocrinology, 14, article 1305606. Available at: https://pubmed.ncbi.nlm.nih.gov/38075079/
Yoo, H.J. et al. (2017) ‘The peptidylglycine α amidating monooxygenase gene rs13175330 A>G polymorphism is associated with hypertension in a Korean population’, Human Genomics, 11, article 24. Available at: https://pubmed.ncbi.nlm.nih.gov/29162152/
Merkler, D.J. et al. (2022) ‘Peptidylglycine α amidating monooxygenase as a therapeutic target or biomarker for human diseases’, British Journal of Pharmacology, 179(13), pp. 3306 to 3324. Available at: https://pubmed.ncbi.nlm.nih.gov/35124797/
Kapuscinski, M. et al. (1993) ‘Peptide alpha amidation activity in human plasma: relationship to gastrin processing’, Clinical Endocrinology, 39(1), pp. 51 to 58. Available at: https://pubmed.ncbi.nlm.nih.gov/8102327/
Wand, G.S. et al. (1985) ‘Characterization of a peptide alpha amidation activity in human plasma and tissues’, Metabolism, 34(11), pp. 1044 to 1052. Available at: https://pubmed.ncbi.nlm.nih.gov/4058309/
Prohaska, J.R. et al. (1997) ‘In vitro copper stimulation of plasma peptidylglycine α amidating monooxygenase in Menkes disease variant with occipital horns’, Pediatric Research, 42(6), pp. 862 to 865. Available at: https://pubmed.ncbi.nlm.nih.gov/9396570/
Maltèse, J.Y. et al. (1990) ‘Characterization of an alpha amidating activity in a human pancreatic tumour secreting vasoactive intestinal peptide’, Clinical Endocrinology, 33(4), pp. 467 to 480. Available at: https://pubmed.ncbi.nlm.nih.gov/2171816/